|
The further study is necessary for obtaining the optimized operation parameters of the IHX to minimize entropy generation and enhance heat transfer. 
With the increasing inlet mass flow rate or LBE inlet temperature, the entropy generation number increases and the heat transfer is enhanced, while the opposite trend occurs with the increasing helium gas inlet temperature. Compared with the results at two working conditions of the constant mass flow rates of liquid lead-bismuth eutectic (LBE) and helium gas, the total pumping power all tends to reduce with the decreasing entropy generation number, but the variations of the effectiveness, number of transfer units and thermal capacity rate ratio are inconsistent, and need to analyze respectively. The variation of entropy generation number with performance parameters of the IHX is analyzed, and effects of inlet conditions of the IHX on entropy generation number and heat transfer are discussed. In the present work, heat transfer and entropy generation of an intermediate heat exchanger (IHX) in the accelerator driven subcritical system (ADS) are investigated experimentally. The intermediate heat exchanger for enhancement heat transfer is the important equipment in the usage of nuclear energy. 
Heat Transfer and Entropy Generation Analysis of an Intermediate Heat Exchanger in ADS The excess entropy of mixing, Δ S ex, at 298 K reaches positive values of 3-4 J mol-1 K-1 in the F-rich portion of the binary, is, however, not significantly different from zero across the join within its 2 σ-uncertainty. They are significant at these conditions exceeding the 2 σ-uncertainty of the data. The heat capacity of the end-members above 298 K can be represented by the polynomials: C, occur in the chlorapatite-fluorapatite solid solution around 80 K (and to a lesser degree at 200 K) and are asymmetrically distributed over the join reaching a maximum of 1.3 ± 0.3 J mol-1 K-1 for F-rich compositions. In such cases, where the sample coupling was not optimal, an empirical procedure was developed to smoothly connect the PPMS to the DSC heat capacities around ambient T. However, the crystals were too small for the crystal face to be polished. Most of the chlor-fluorapatite compositions could be measured directly as single crystals using the PPMS such that they were attached to the sample platform of the calorimeter by a crystal face. The chlor-fluorapatites were synthesized at 1,375-1,220Â☌ from Ca3(PO4)2 using the CaF2-CaCl2 flux method. The heat capacity at constant pressure, C p, of chlorapatite, and fluorapatite, as well as of 12 compositions along the chlorapatite-fluorapatite join have been measured using relaxation calorimetry and differential scanning calorimetry (DSC) in the temperature range 5-764 K. From the equilibrium data of Bowen and Tuttle and the entropy of talc, the heat of formation of talc from MgO, SiO2, and H2O (liq.) is calculated to be ?H?f298 = -43.6 ? 1 kcal.Įxcess heat capacity and entropy of mixing along the chlorapatite-fluorapatite binary joinĭachs, Edgar Harlov, Daniel Benisek, Artur the values of the thermodynamic functions are: talc, Cp? = 76.89 ? 0.23 cal. Smoothed values of heat capacity, entropy, enthalpy, and free energy are tabulated. The heat capacities of talc, Mg3Si4O10(OH)2, and tremolite, Ca2Mg6Si8O22(OH)2, have been measured between 12 and 305?K. Heat capacity from 12 to 305°K and entropy of talc and tremolite The uncertainties are twice the standard deviation of the mean. At T = 298.15 K, the recommended values for the molar heat capacity, molar entropy, and molar enthalpy increment referred to T = 0 are (25.77 ? 0.08) J?K-1?mol-1, (42.90 ? 0.11) J?K-1?mol-1, and (5811 ? 12) J?mol-1, respectively. 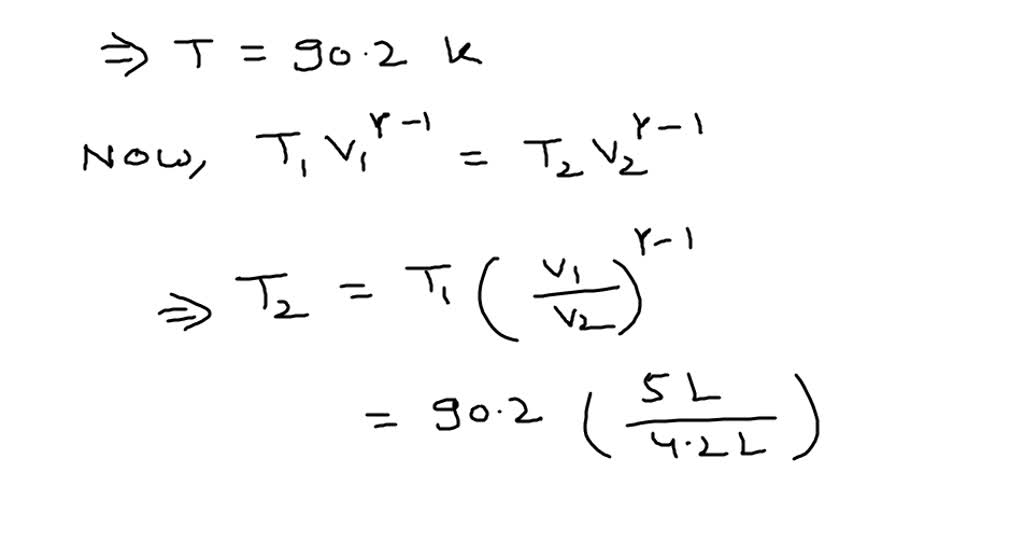
The heat capacity of calcium has been measured at 85 mean temperatures between T ? 8 K and T ? 369 K using an adiabatically-shielded calorimeter in an intermittent heating mode. Molar heat capacity and entropy of calcium metal
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
